|
Another state of matter is Bose-Einstein condensate, but this state only occurs with super-low temperatures. One state is plasma, which naturally occurs in lightning, and we create it in fluorescent light bulbs and plasma TVs. We’ve known about solids, liquids and gases for hundreds of years, but scientists have discovered other states. We cannot see the water vapour as we exhale, but if we hold our eyeglasses or smartphone to our mouths, we can see the water vapour condensing (becoming liquid) on these objects. We exhale water vapour whenever we breathe out. Students will understand that matter can be found in three forms or phases (solid, liquid, and gas). Unlike ice or water, water vapour is invisible. of water molecules in the three physical states. Water vapour is the gaseous form or state of water. (Think about a diving tank – 600 L of gas is compressed into a 3 L cylinder.) Right now, you are breathing in air – a mixture of gases containing many elements such as oxygen and nitrogen. Gas can be compressed much more easily than a liquid or solid. A gas will fill any container, but if the container is not sealed, the gas will escape. They vibrate and move freely at high speeds. The atoms and molecules in gases are much more spread out than in solids or liquids. Anaheim (Ca) USA.Find out more about water by looking at our wide range of resources under the water topic. (1986) Improved Durand-equation for multiple application. (1978) Pseudohomogene Gemische, Teil B, aus: Hydraulischer Feststofftransport in Rohrleitungen. Minimum transport velocity for large particle size suspensions in round horizontal pipes. (1962) Transport characteristics of suspensions: Part VI. (1958) A generalized theory of sedimentation. (1953) Basic relationships of the transportation of Solids in pipes-experimental research. (1986) Drag coefficient and settling velocity of particles. Experimental studies on the hydraulic transport of iron ore.ĭedegil, M. (1986) Improved Durand-equation for multiple application. Pure, crystalline solids have a characteristic melting point, the temperature at which the solid melts to become a liquid.The transition between the solid and the liquid is so sharp for small samples of a pure substance that melting points can be measured to 0.1 o C. 
(1977) Solid liquid flow-slurry pipeline transportation. Fluid velocities required for this condition can be obtained from Table 1.ĭedegil, M. To guarantee equal conditions for solids of any density relative to the concentration profile at these Re s limits, the ratio of the settling velocity and the fluid velocity w so/u L must remain constant. 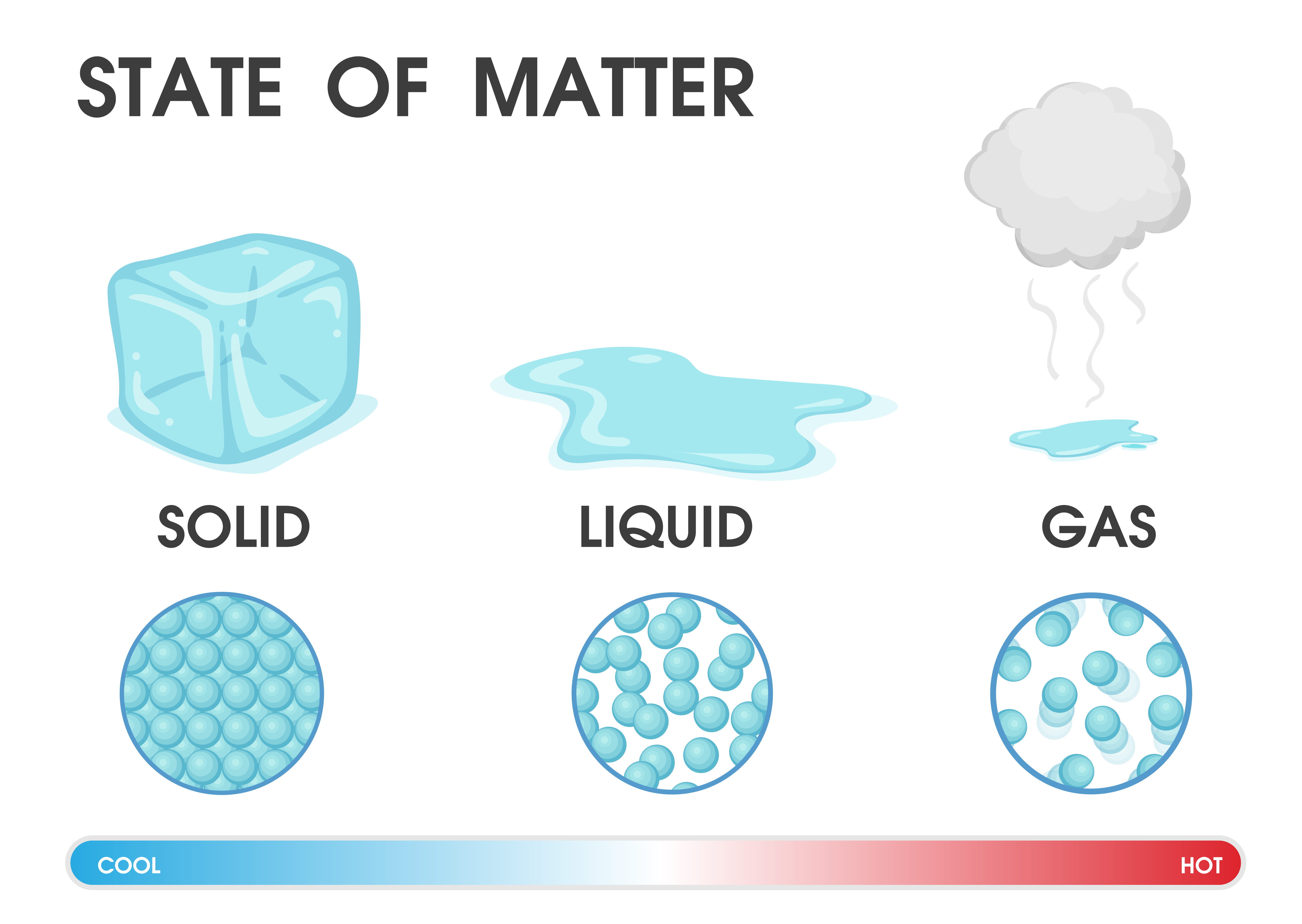
This type of suspension can exist at economically-feasible velocities and is called the pseudo-homogeneous flow regime. 
A certain degree of segregation is permitted. 
Particles with 0.1 < Re s < 2 need some more turbulence and velocity to be held in suspension, but in the case of horizontal flow, completely uniform solid distribution cannot be reached. Only little turbulence is needed to keep those particles homogeneously suspended so liquid velocity can be low in this homogeneous flow regime. Only very fine particles with Re s <10 −6, which can be conveyed by Brownian Molecular Movement, are kept in suspension without any Turbulence, the so-called colloidal dispersions.įine particles with 10 −6 < Re s < 0.1 can be easily held in suspension by hydraulic forces, and this tendency is supported by a low solid density and by a nonspherical particle shape.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
